 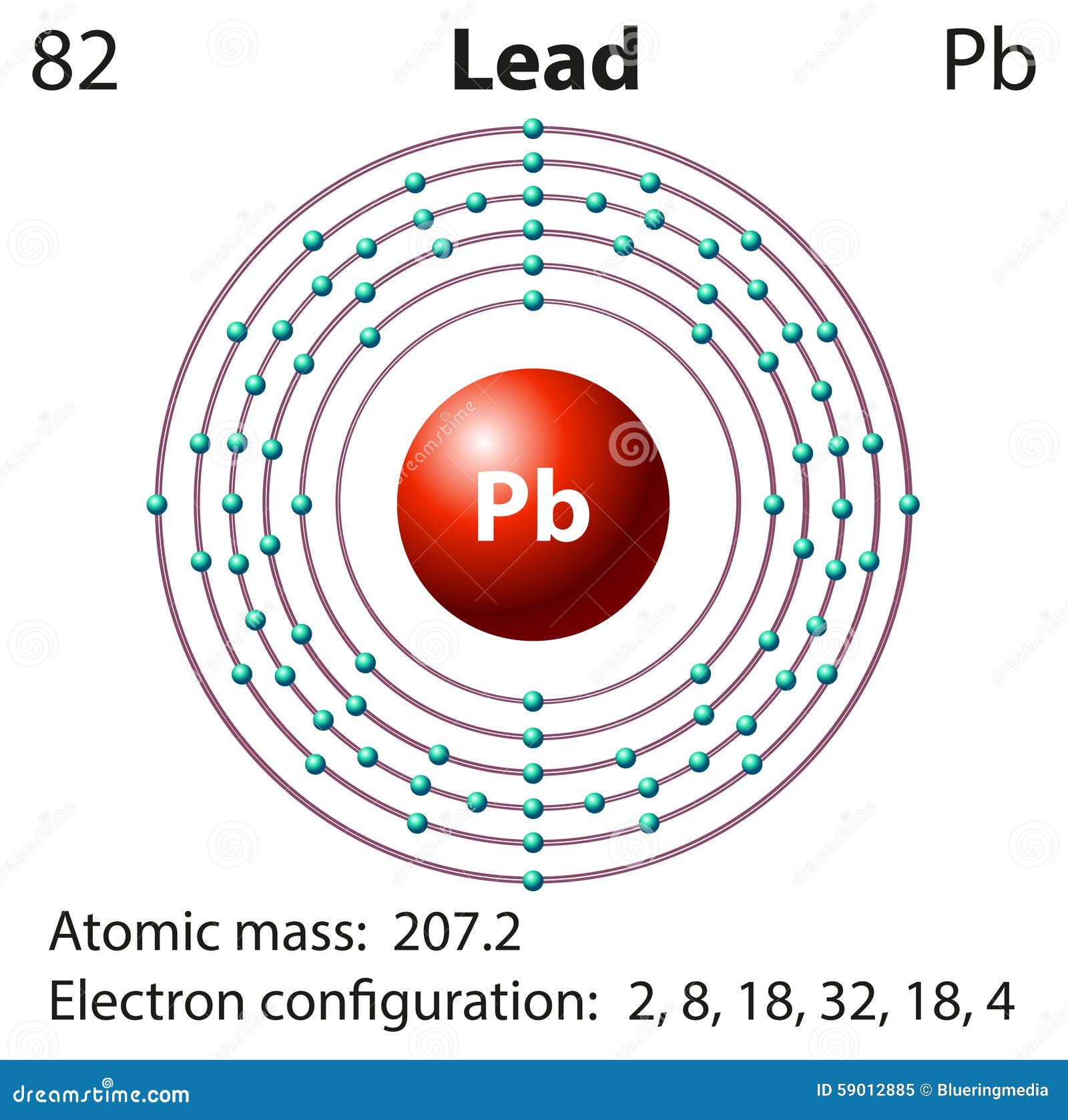
Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. 
These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. 
Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. This study has implications for Pb/Pb geochronological data particularly in lower grade metamorphic terrains.A vertical column in the periodic table. (BNL), Upton, NY (United States) Sponsoring Org.: USDOE Office of Science (SC), Nuclear Physics (NP) International Union of Pure and Applied Chemistry (IUPAC) OSTI Identifier: 1615597 Report Number(s): BNL-213840-2020-JAAM Journal ID: ISSN 1365-3075 TRN: US2106368 Grant/Contract Number: SC0012704 Resource Type: Journal Article: Accepted Manuscript Journal Name: Pure and Applied Chemistry (Online) Additional Journal Information: Journal Volume: 93 Journal Issue: 1 Journal ID: ISSN 1365-3075 Publisher: de Gruyter Country of Publication: United States Language: English Subject: 73 NUCLEAR PHYSICS AND RADIATION PHYSICS atomic-weight interval atomic weights geochronology isotopic abundances IUPAC Inorganic Chemistry Division radioactive radiogenic standard atomic = 1,700 Ma Laxfordian resetting of their Pb-isotope systematics. Publication Date: Research Org.: Brookhaven National Lab. US Geological Survey, Reston, VA (United States).Chinese Academy of Sciences (CAS), Beijing (China).It is proposed that a value of 207.2 be adopted for the single lead atomic-weight value for education, commerce, and industry, corresponding to previously published conventional atomic-weight values. When expressed as an interval, the lead atomic weight is. The highest published lead atomic weight is 207.9351 ± 0.0005 (k = 2) for monazite from a micro-inclusion in a garnet relic, also from a high-grade metamorphic more » terrain in north-western Scotland, which contains almost pure radiogenic 208Pb. 
In a comprehensive review of several hundred publications and analyses of more than 8000 samples, published isotope data indicate that the lowest reported lead atomic weight of a normal terrestrial materials is 206.1462 ± 0.0028 (k = 2), determined for a growth of the phosphate mineral monazite around a garnet relic from an Archean high-grade metamorphic terrain in north-western Scotland, which contains mostly 206Pb and almost no 204Pb. While elemental lead can serve as an abundant and homogeneous isotopic reference, deviations from the isotope ratios in other lead occurrences limit the accuracy with which a standard atomic weight can be given for lead. These variations in isotope ratios and atomic weights provide useful information in many areas of science, including geochronology, archaeology, environmental studies, and forensic science. The lightest stable isotope, 204Pb, is primordial. The isotopic composition and atomic weight of lead are variable in terrestrial materials because its three heaviest stable isotopes are stable end-products of the radioactive decay of uranium ( 238U to 206Pb 235U to 207Pb) and thorium ( 232Th to 208Pb). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
